Education Requirements
All investigators must comply with Woman's Hospital's Human Research Protection Education and Training Requirements Policy.
You must have human subjects research training to participate in research activities at Woman’s.
If you are a Woman’s employee or affiliate, you must have completed the CITI Human Subjects Research certification. CITI HSR certifications last for 3 years, can be renewed as needed, and are available at no cost to you. If you are conducting a clinical trial, you must also have the CITI Good Clinical Practice certification. If you are affiliated with an outside organization, you may provide the respective human subjects protections training certificates required by your organization.
Learn how to register for CITI training here.
CITI Training for Research Involving Human Subjects:
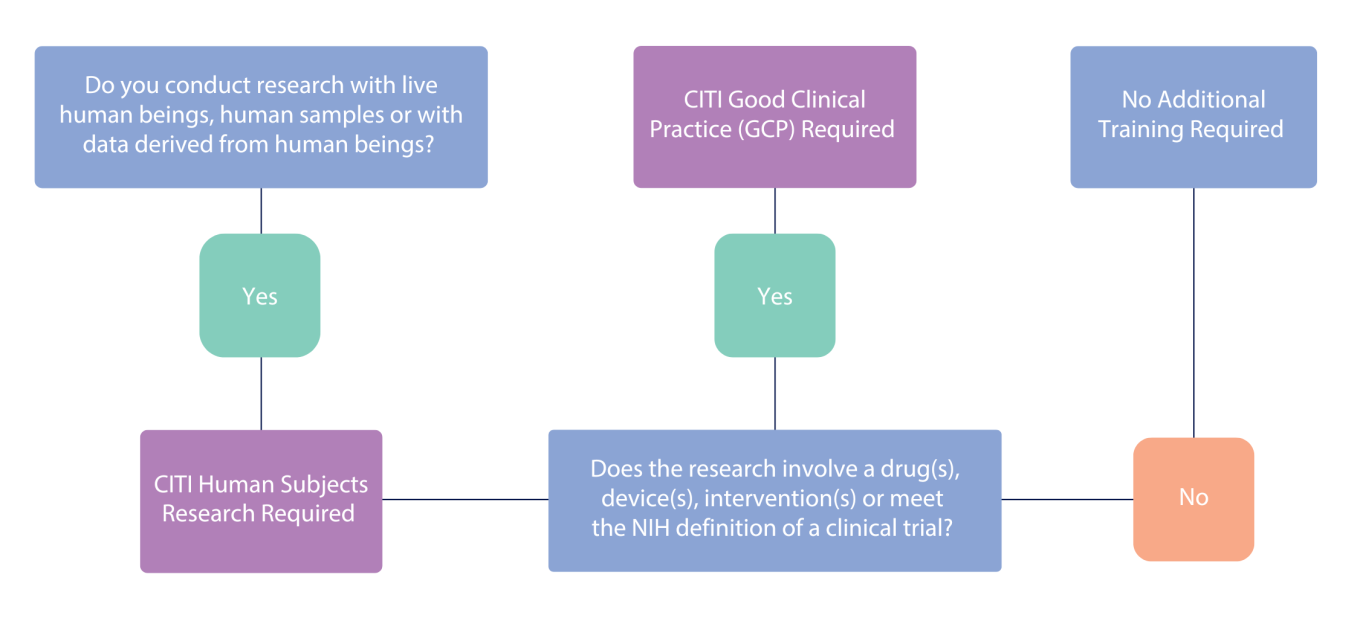
Definitions:
- Clinical Trial: A research study in which one or more human subjects are prospectively assigned to one or more interventions (which may include placebo or other control) to evaluate the effects of those interventions on health-related biomedical or behavioral outcomes.
- Intervention: A manipulation of the subject or subject’s environment for the purpose of modifying one or more health-related biomedical or behavioral processes and/or endpoints. (e.g., drugs/small molecules/compounds; biologics; devices; procedures (e.g., surgical techniques); delivery systems (e.g., telemedicine, face-to-face interviews); strategies to change health-related behavior (e.g., diet, cognitive therapy, exercise, development of new habits); treatment strategies; prevention strategies; and, diagnostic strategies.
- Investigator: The individual responsible for the conduct of the clinical trial at a trial site. (e.g., Principal Investigator).
- Clinical trial staff: Individuals, identified by the investigator, who are responsible for study coordination, data collection, and data management. The central focus of clinical trial staff is to manage participant recruitment and enrollment, to maintain consistent study implementation, data management, and to ensure integrity and compliance with regulatory and reporting requirements. These individuals may also seek informed consent from prospective participants, enroll and meet with research participants, and collect and record information from research participants. Clinical trial staff may also be called the research coordinator, study coordinator, research nurse, study nurse, or subinvestigator.